|
11/8/2023 0 Comments Electron domain geometry for brf3The noble gas xenon difluoride is a hypervalent halogen compound with an octet rule exception and no net dipole moment. As a result, the XeF 2 molecule’s hybridisation is sp 3d. As a result, the core atom Xe is sp 3d hybridised. 
Xenon’s electron configuration changes to 5s 2 5p 5 5d 1 with two unpaired electrons. On the other hand, in XeF 2, the Xe molecule is excited. The xenon’s ground state comprises eight electrons grouped in 5s 2 5p 6 orbitals. The outer shell of Xenon in XeF 2 includes eight electrons, two of which are involved in bond formation. Two or more orbitals with differing energy levels combine to generate hybrid orbitals during bond formation. Understanding the geometry of a specific molecule necessitates hybridisation. To create a near-stable composite, this idea minimises the like charge repulsion between negative electron clouds around atomic nuclei. To compute the exact molecular structure and bond lengths and angles, we use a theory called the Valence Shell Electron Pair Repulsion Model (VSEPR). This is a shift from 2D to 3D structural representation, which allows us to see how a molecule stays in a bonding state in real life. If we want to learn more about how a molecule’s interior appears, we must research molecular geometry. The molecular geometry of XeF 2 is linear because the bond angle between the two pairs coupled with the centre atom is 180 degrees. 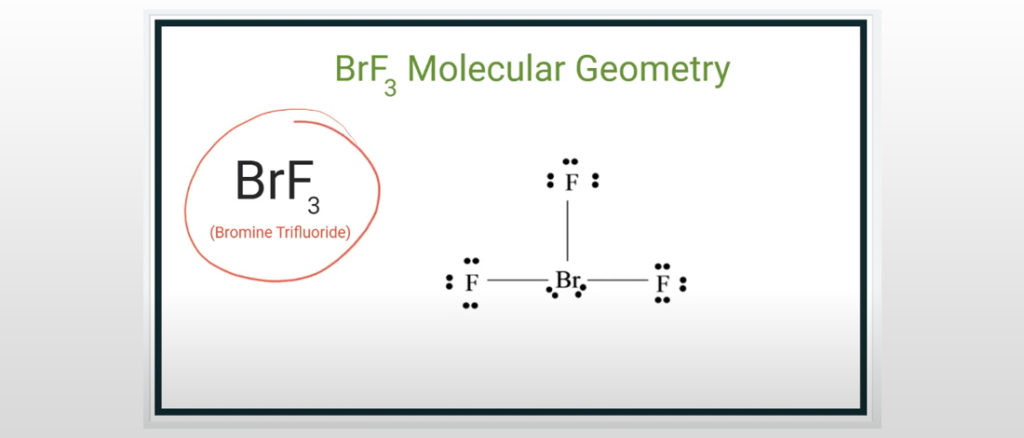
Compared to the bond pairs, the lone pairs are in an equatorial location. There are three lone pairs of electrons and two pairs of bond electrons. The bond angle may be easily comprehended now that we know the chemical geometry of the Xenon Difluoride molecule. Carbon, for example, belongs to group 4 (also known as Group XIV) and hence contains four electrons in its valence shell. The groups will help us figure out what an atomic element’s valence electron number is when looking at the periodic table. Valence electrons are the electrons in an atom’s outermost shell that control its valency. The notion of valence electrons is the first thing to comprehend about chemical bonding. Straight lines reflect the bond formation in the molecule, whereas dots represent the lone pairs. Both bonded and lone pairs of electrons are depicted differently to distinguish between the two types of electrons. Lone pairs of electrons are those that do not participate in bond formation. The electrons that participate in bond formation and those that do not are referred to as valence electrons collectively.īonding pairs of electrons are electrons that participate in the formation of bonds. This structure aids in deciphering the charges on the compound’s molecules. It is a visual representation of all the electrons involved in bond formation. The Lewis structure of a chemical and its molecular geometry is important for understanding all of its physical and chemical properties. XeF 2 Molecular Geometry: Lewis Structure The XeF 2 molecular geometry and bond angles have a lot of importance, so there are a few words and ideas that you should be familiar with before studying XeF 2 molecular geometry and bond angles notes. Chemical bonding is the backbone that defines a molecule complex’s internal structure and nature and characteristics (both physical and chemical). Molecules are generated through the formation of certain bonds between atoms that are based on their strength. Homogeneous and heterogeneous molecules are formed when two or more atoms react and combine. 
XeF 2 Molecular Geometry: Chemical Adhesion It is left with three lone pairs placed in the equatorial plane and two fluorines arranged perpendicularly to the lone pairs in the axial plane after sharing 1–1 electron with fluorine. Due to the 180-degree bond angles, the structure must be linear. Although the lone pairs are at equatorial positions (bonds perpendicular to the axis), the molecule is a trigonal bipyramid. This indicates that both fluorines must be bound to the Xe molecule, resulting in three unshared pairs and two bonded pairs on the Xe molecule. Molecule Xe has eight valence electrons, while fluorine has seven, totalling 22 valence electrons. XeF 2 molecular geometry is an important and interesting topic. XeF 2 is generated when a 2:1 mixture of xenon and fluorine is heated at 400 ℃ in a sealed nickel tank. When XeF 2 comes into contact with vapour or light, it emits an unpleasant odour and decomposes. Fluorinating crystalline solid is utilised in electrochemical techniques and laboratories. XeF 2 is the most stable of the three chemicals. Other Xenon compounds include XeF 4 (Xenon Tetrafluoride) and XeF 6 ( Xenon Hexafluoride). It’s a fluorinating and oxidising agent with a lot of punch. The chemical compound Xenon Difluoride is abbreviated as XeF 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
